“I hope that one day we’ll know where we are and how many of us there are.”
Joanda Gomes, Pernambuco, Brasil
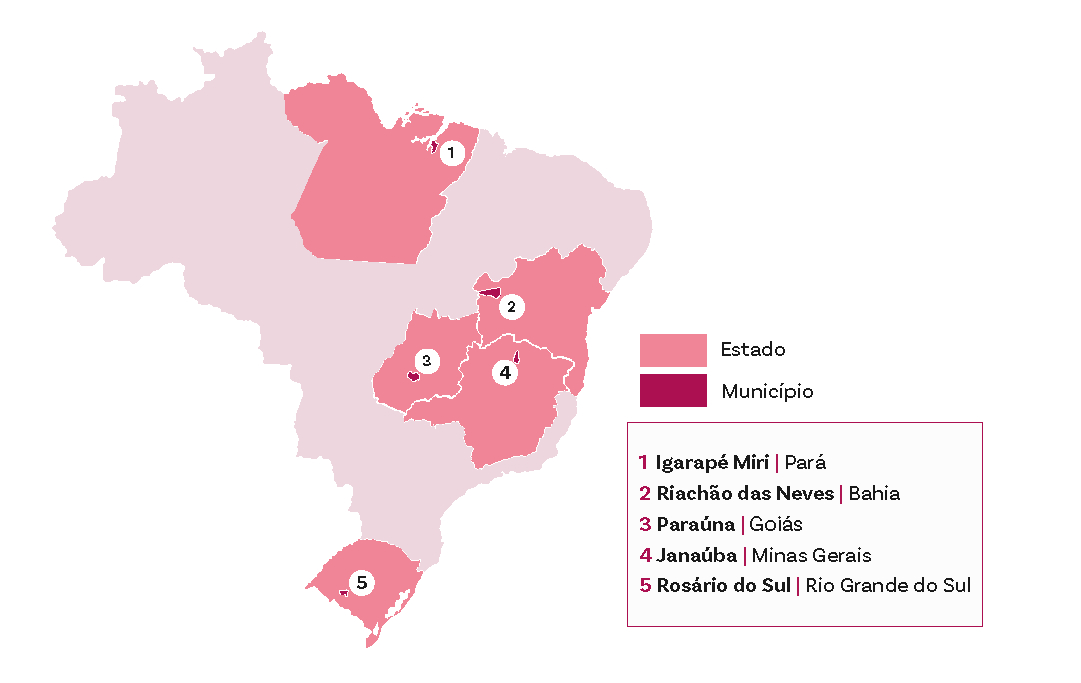
In Brazil, despite advances in vector control, Chagas’ disease remains a persistent public health challenge. Estimates from the Ministry of Health indicate a prevalence of 1.0% to 2.4% of the population, which equates to approximately 1.9 to 4.6 million people infected with Trypanosoma cruzi.
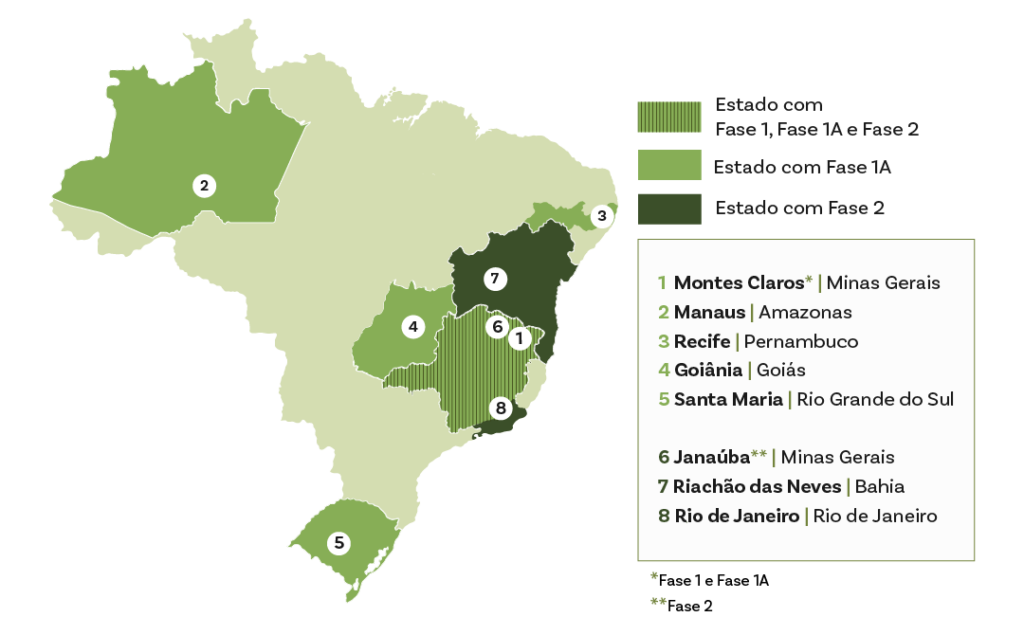
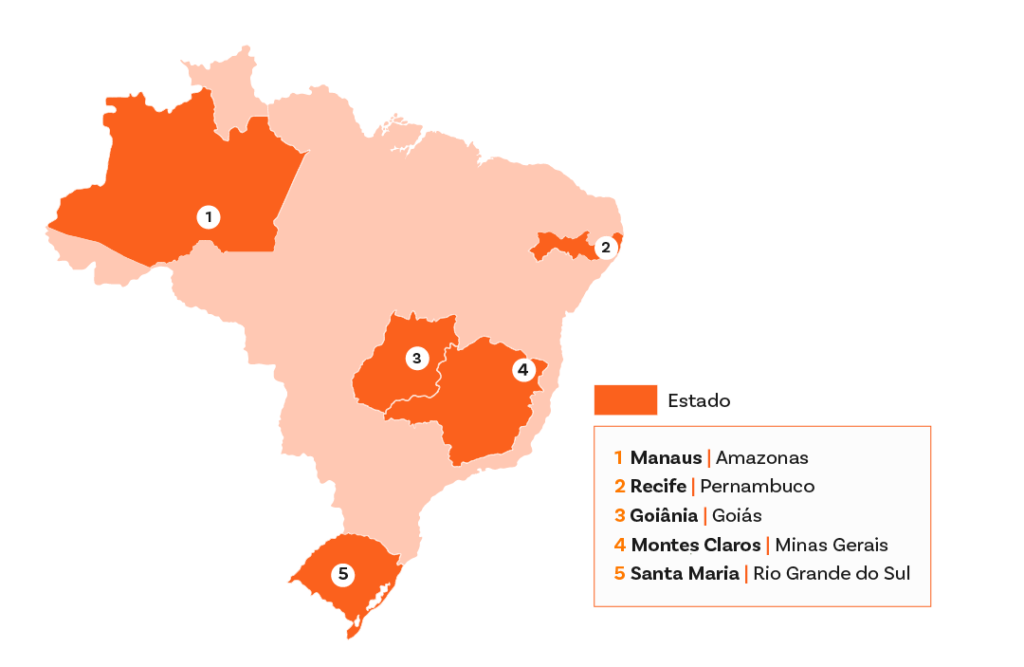
The disease continues to have a high mortality rate in the country, and acute transmission still occurs in various states, with a recent increase in the Northern region. Within the CUIDA Chagas initiative, activities in Brazil integrate diagnosis, care and community participation, linking primary care, specialist services and local partners.
Studies in Brazil
Validation study of a diagnostic algorithm using rapid tests
News from Brazil
Oswaldo Cruz Foundation (Fiocruz)
4036 Avenida Brasil, Room 102 – Manguinhos – Rio de Janeiro, RJ, Brazil – Postcode: 21040-361 – info@cuidachagas.org